In early mammalian development, oocytes accumulate large amounts of maternal proteins and RNAs prior to maturation, providing the molecular foundation for the maternal-to-zygotic transition (MZT) and subsequent zygotic genome activation (ZGA) after fertilization. Maternal effect genes (MEGs) are expressed in oocytes, of which the products are not necessarily required during oocyte maturation, but become indispensable during embryonic development after fertilization. Previously identified MEGs have largely been linked to epigenetic regulation or RNA metabolism. Because the first round of DNA replication in early embryos typically takes place before embryo genome activation when transcription is initiated, maternal factors are crucial for ensuring the successful completion of the embryo’s first DNA replication.
On 23 February 2026, Prof. Wei Xie (School of Life Sciences, Tsinghua University) and Prof. Shuyan Ji (Zhejiang University School of Medicine; Institute of Medical Genetics and Development) reported their collaborative study in EMBO Reports as a Research Article entitled “THAP1 is a maternal effect factor required for the first cell cycle via Rrm1 in early mouse embryos.” This study demonstrates that the transcription factor THAP1 is highly expressed in oocytes and activates a small but critical set of genes, including Rrm1. RRM1 contributes to dNTP production, and its accumulation in oocytes ensures the successful first DNA replication after fertilization. When maternal THAP1 is absent, embryos show a pronounced delay in 1-cell DNA replication, frequent 1–2-cell arrest, and ZGA defects, ultimately leading to a severe reduction in female fertility.
THAP1 is a THAP-domain transcription factor of the Thanatos-associated protein family. In human, mutations in THAP1 can cause a neurological movement disorder, dystonia type 6 (DYT6) (Fuchs et al., 2009). Compared to differentiated tissues, THAP1 is strongly expressed in oocytes and preimplantation embryos; however, its specific function during early embryogenesis remained unexplored. To address this question, the research team generated an oocyte-specific maternal Thap1 knockout mouse model (Thap1 mKO). The results showed that loss of maternal Thap1 caused no obvious abnormalities in oocyte growth or meiotic maturation. However, embryonic development after fertilization from the 1-cell to the 2-cell stage was significantly delayed, with many embryos stalled at the 1–2-cell stage and exhibiting ZGA impairment, ultimately resulting in markedly reduced female reproductive capacity. Increased DNA damage, sustained activation of cell-cycle checkpoints, and delayed S phase exit was observed during DNA replication in the 1-cell embryos, echoing the defects of 1-cell to 2-cell transition.
To investigate the underlying mechanisms, the team conducted transcriptome profiling and found that Thap1 deficiency led to downregulation of a small subset of genes in oocytes, including Rrm1, which encodes a subunit of ribonucleotide reductase. RRM1 is required for dNTP biosynthesis, a core prerequisite for DNA replication. To determine whether Thap1 loss indeed affects nucleotide availability, the researchers used an optimized, label-free HPLC–MS strategy to map the metabolomes spanning from the oocyte growth to the zygote stages. Indeed, dNTP levels were reduced in Thap1 mKO oocytes. Remarkably, overexpression of Rrm1 alone in zygotes nearly fully rescued the 2-cell transition and ZGA. Supplementation with dNDPs (the catalytic products of ribonucleotide reductase) also partially alleviated the defects in Thap1 mKO embryos. Together, these findings support an essential role of the “THAP1–Rrm1–dNTP supply” in the first cell cycle following fertilization.
In summary, this work establishes THAP1 as a maternal-effect factor in early mouse development by safeguarding the first DNA replication of life through transcribing nucleotide-metabolism genes in oocytes (Fig. 1).
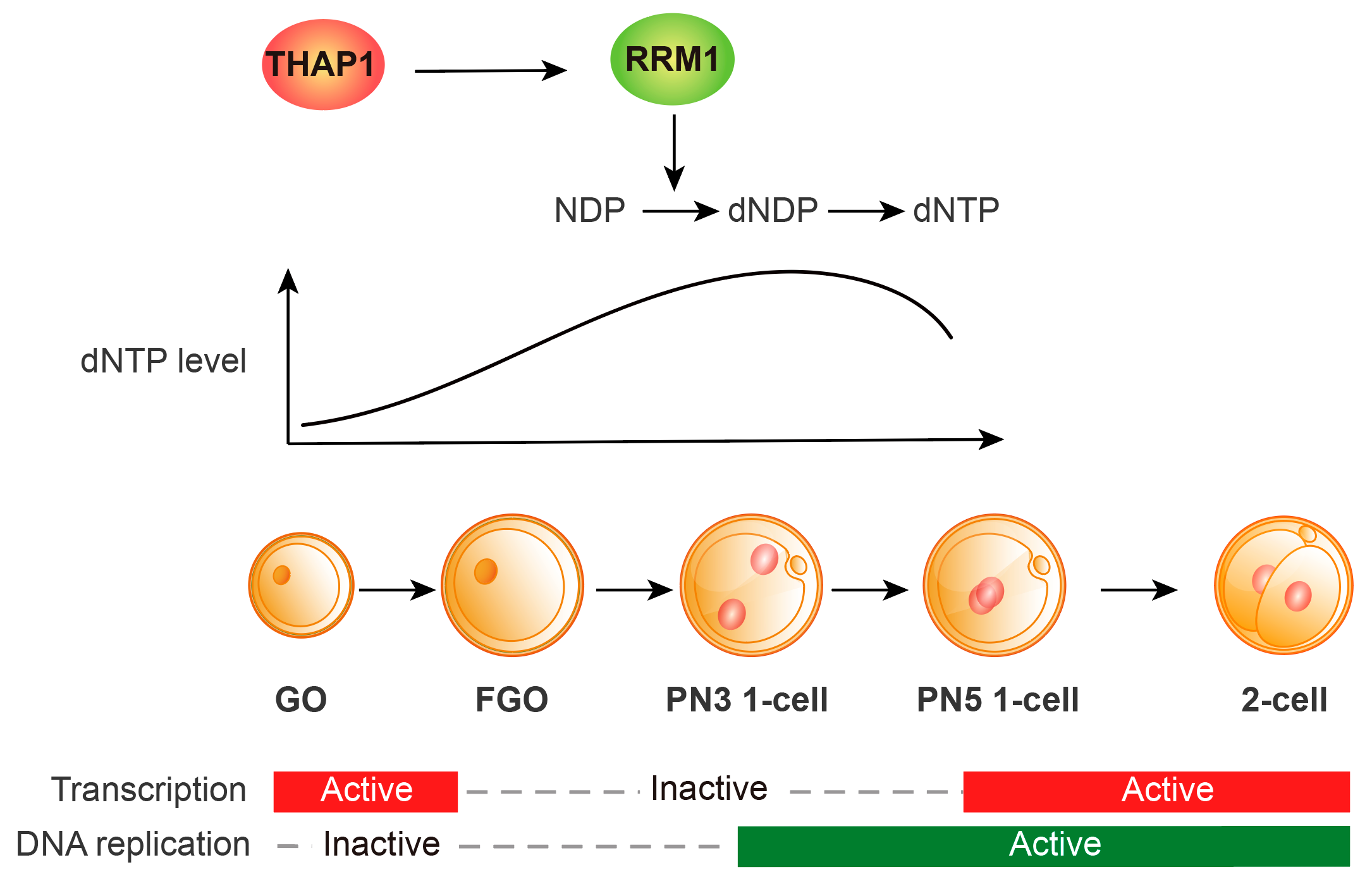
Fig 1. THAP1 ensures adequate dNTP supply for the first cell cycle after fertilization by transcriptionally activating the downstream gene Rrm1, thereby supporting the 1-cell to 2-cell transition and ZGA.
Prof. Wei Xie (Tsinghua University School of Life Sciences) and Prof. Shuyan Ji (Zhejiang University School of Medicine, Institute of Medical Genetics and Development) (a formal postdoctoral fellow in Wei Xie lab) are the corresponding authors of the paper. Qiang Fan (Ph.D. student, Tsinghua University), Xi Wu (Ph.D. student, PTN Program, Peking University), and Yanna Dang (postdoctoral fellow, Tsinghua–Peking University Center for Life Sciences) are the co-first authors. The study was supported by the Tsinghua University Laboratory Animal Center and the Tsinghua University Bio-computing Center, and funded by the National Key R&D Program of China, the National Natural Science Foundation of China, and the Tsinghua–Peking University Center for Life Sciences. Wei Xie is a New Cornerstone Investigator.
Paper link: https://doi.org/10.1038/s44319-026-00712-9
