A collaborative work by Dieter Wolf’s group at Xiamen University and Xuerui Yang’s group at Tsinghua has led to a new research article, “eIF3 Associates with 80S Ribosomes to Promote Translation Elongation, Mitochondrial Homeostasis, and Muscle Health”, which was recently published in Molecular Cell. Link to the paper: https://www.cell.com/molecular-cell/fulltext/S1097-2765(20)30388-9.
Protein synthesis by mRNA translation is a highly coordinated process, which is essential for many fundamental physiological functions. An extensive array of eukaryotic initiation factors (eIFs) and additional ribosome-associated proteins take part in the regulation of translation at different stages. eIF3 is a canonical regulator of translation, which has been believed to function during initiation.
Years ago, Dieter Wolf’s research group at Xiamen University and Xuerui Yang’s group at Tsinghua teamed up with an aim to dissect the exact functions of eIF3 in translation, given some preliminary information that seemed inconsistent to the canonical theory about the protein eIF3. The two groups developed a series of experiments and new strategies for mining of the multi-omics data of the transcriptome, translatome, and proteome upon perturbation of eIF3. The joint efforts led to a totally unexpected finding that eIF3 deficiency reduced early ribosomal elongation speed between codons 25 and 75 on a set of ~2,700 mRNAs encoding proteins associated with mitochondrial and membrane functions. To promote elongation, eIF3 interacts with 80S ribosomes translating the first ~60 codons and serves to recruit protein quality-control factors, functions required for normal mitochondrial physiology. Accordingly, eIF3e knock-out mice accumulate defective mitochondria in skeletal muscle and show a progressive decline in muscle strength. Hence, eIF3 interacts with 80S ribosomes to enhance, at the level of early elongation, the synthesis of proteins with membrane-associated functions, an activity that is critical for mitochondrial physiology and muscle health.
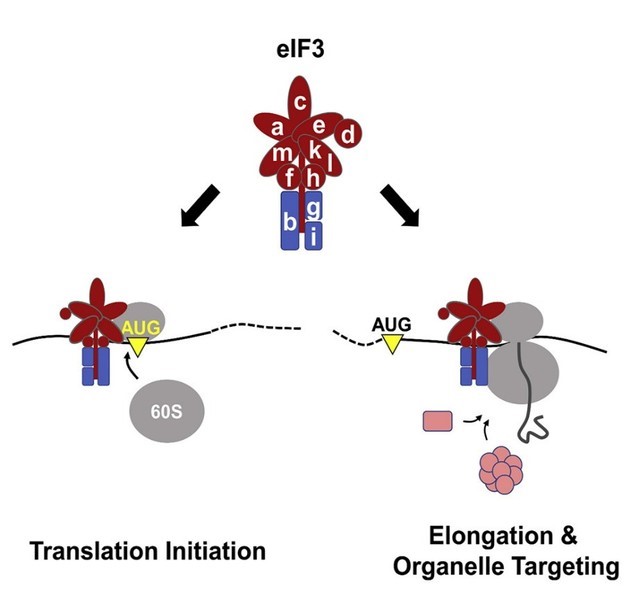
Schematic diagram showing the functional model of eIF3 at different stages of translation
In summary, the work has uncovered a hidden role of eIF3 in regulating translation of a specific group of genes encoding mitochondria and membrane proteins. This new discovery painted potential links between eIF3 and complex diseases related to the dysfunctional mitochondria, such as cancer and neurodegenerative diseases.
The work has been done by a collaborative team from Dieter Wolf’s lab at Xiamen University and Xuerui Yang’s lab at Tsinghua. The research has greatly benefitted from Wolf’s mechanistic study of translation regulation and Yang’s methodology in mining of the translatome data. Dieter Wolf and Yabin Cheng at Xiamen and Xuerui Yang at Tsinghua serve as co-corresponding authors. Yingying Lin at Xiamen and Fajin Li at Tsinghua are co-first authors with equal contributions. Xuerui Yang’s work was financially supported by the national key research and development program, Precision Medicine Project, the National Natural Science Foundation of China, the Tsinghua University Initiative Scientific Research Program. The study received supports from the core facilities of Genome Sequencing and High-Performance Computing of the National Protein Science Facility (Beijing).
